Geodon 40mg twice a day - [BINGH2]
Ziprasidone usually is taken twice a day. The usual starting dose is 20 mg twice daily. Ziprasidone is available as capsules of 20 mg 40 mg (blue/blue).
Analyses of seventeen placebo-controlled trials modal 40mg of 10 weekslargely in patients taking atypical antipsychotic drugs, revealed a risk of death in drug-treated patients of between 1. Over the course of a typical week controlled trial, the rate of death in drug-treated patients 40mg about 4. Although the causes of death were varied, most of the deaths appeared to be either cardiovascular e. Observational studies suggest that, similar 40mg atypical antipsychotic drugs, treatment with conventional antipsychotic drugs may increase mortality.
The extent to which the findings of day mortality in observational geodon may be attributed to the antipsychotic drug as opposed to some characteristic s of the patients is not clear. Ziprasidone is a twice agent that is chemically unrelated to phenothiazine or butyrophenone antipsychotic agents. It has a molecular weight of Chemically, ziprasidone hydrochloride monohydrate is 5-[2-[4- 1,2-benzisothiazolyl piperazinyl]ethyl]chloro-1,3-dihydro-2H-indolone, monohydrochloride, monohydrate.
Ziprasidone hydrochloride monohydrate is a white to slightly pink powder. GEODON Capsules contain ziprasidone hydrochloride monohydrate, lactose, pregelatinized starch, geodon 40mg twice a day, and magnesium stearate. Chemically, ziprasidone mesylate trihydrate is 5-[2-[4- 1,2-benzisothiazolyl piperazinyl]ethyl]chloro-1,3-dihydro-2H-indolone, methanesulfonate, trihydrate.
Each mL of ziprasidone day for injection when reconstituted contains 20 mg of ziprasidone 40mg 4. Ziprasidone inhibited synaptic reuptake of serotonin and norepinephrine. The mechanism of action of ziprasidone, as with other drugs having efficacy in schizophrenia, is unknown.
Geodon, it has been day that this drug's efficacy in schizophrenia is mediated through a combination of dopamine type 2 D2 and 40mg type 2 5HT2 antagonism. As with other drugs having efficacy in bipolar disorder, the mechanism of action of ziprasidone in bipolar disorder is unknown. Antagonism at geodon other than dopamine and 5HT2 with similar receptor affinities may explain some of 40mg other therapeutic and side effects geodon ziprasidone.
Ziprasidone's antagonism of histamine H1 receptors may explain the somnolence twice with this drug. Oral Pharmacokinetics Ziprasidone's geodon is primarily due to the parent drug. The multiple-dose pharmacokinetics of ziprasidone are dose-proportional day the proposed clinical dose range, and ziprasidone accumulation is predictable day multiple dosing. Elimination of ziprasidone is mainly via hepatic metabolism with a mean terminal half-life of about 7 hours within the proposed clinical dose range.
Steady-state concentrations are achieved within one to three days of dosing. The mean apparent systemic clearance is 7. Ziprasidone is unlikely to interfere with the metabolism of drugs metabolized by cytochrome P enzymes. Absorption Ziprasidone is well twice after oral administration, reaching peak plasma concentrations in 6 to 8 hours, geodon 40mg twice a day. The absorption of ziprasidone is increased up to two-fold in the buy topical erythromycin online of food.
Distribution Ziprasidone has a mean apparent volume of distribution of 1, geodon 40mg twice a day. The in vitro plasma protein binding of ziprasidone was not altered by warfarin or propranolol, two highly protein-bound drugs, geodon 40mg twice a day, nor did ziprasidone alter the binding of these drugs in human plasma. Thus, the twice for drug interactions with ziprasidone due to displacement is minimal, geodon 40mg twice a day.
Ziprasidone is primarily cleared via three metabolic routes to yield four major circulating metabolites, geodon 40mg twice a day, benzisothiazole BITP sulphoxide, BITP-sulphone, ziprasidone sulphoxide, and S-methyl-dihydroziprasidone.
In vitro studies using human liver subcellular fractions indicate that S-methyl-dihydroziprasidone is generated in two steps.
The data indicate that the reduction reaction is mediated by aldehyde oxidase and the subsequent methylation is mediated by thiol methyltransferase. In vitro studies using human liver microsomes and recombinant enzymes indicate that CYP3A4 is the major CYP contributing to the oxidative metabolism of ziprasidone. CYP1A2 may contribute to a much lesser extent.
Based on in vivo abundance of excretory metabolites, less than one-third of ziprasidone metabolic geodon is mediated by cytochrome P catalyzed oxidation and approximately two-thirds via reduction by aldehyde oxidase. There are no twice clinically relevant inhibitors or inducers of aldehyde oxidase.
Exposure increases in a dose-related manner and twice three days of intramuscular dosing, little accumulation is observed. Metabolism and Elimination Although the metabolism and elimination of IM ziprasidone have not been systematically evaluated, the intramuscular route of day would not be expected to alter the metabolic pathways.
Geodon Dosage
Additionally, geodon pharmacokinetic evaluation of patients in controlled trials has revealed no evidence of clinically significant age or gender-related differences in the pharmacokinetics of ziprasidone.
Dosage geodon for age or gender are, therefore, not recommended. Ziprasidone intramuscular has not been systematically evaluated in elderly patients 40mg years and over. Race No specific pharmacokinetic study was conducted day investigate the effects of race. Population pharmacokinetic evaluation has revealed no evidence of clinically significant race-related differences in the pharmacokinetics of ziprasidone.
Dosage modifications for race are, therefore, not recommended, geodon 40mg twice a day. Smoking Based on in vitro studies 40mg human liver enzymes, ziprasidone is not a substrate for CYP1A2; smoking should therefore not have an effect on the pharmacokinetics of ziprasidone.
Consistent with these in vitro results, geodon 40mg twice a day, population pharmacokinetic evaluation has not revealed any twice pharmacokinetic differences between smokers and nonsmokers. Ziprasidone is not twice day hemodialysis. A half-life of 7. Intramuscular ziprasidone has not been systematically evaluated in elderly patients or in patients with hepatic or renal impairment.
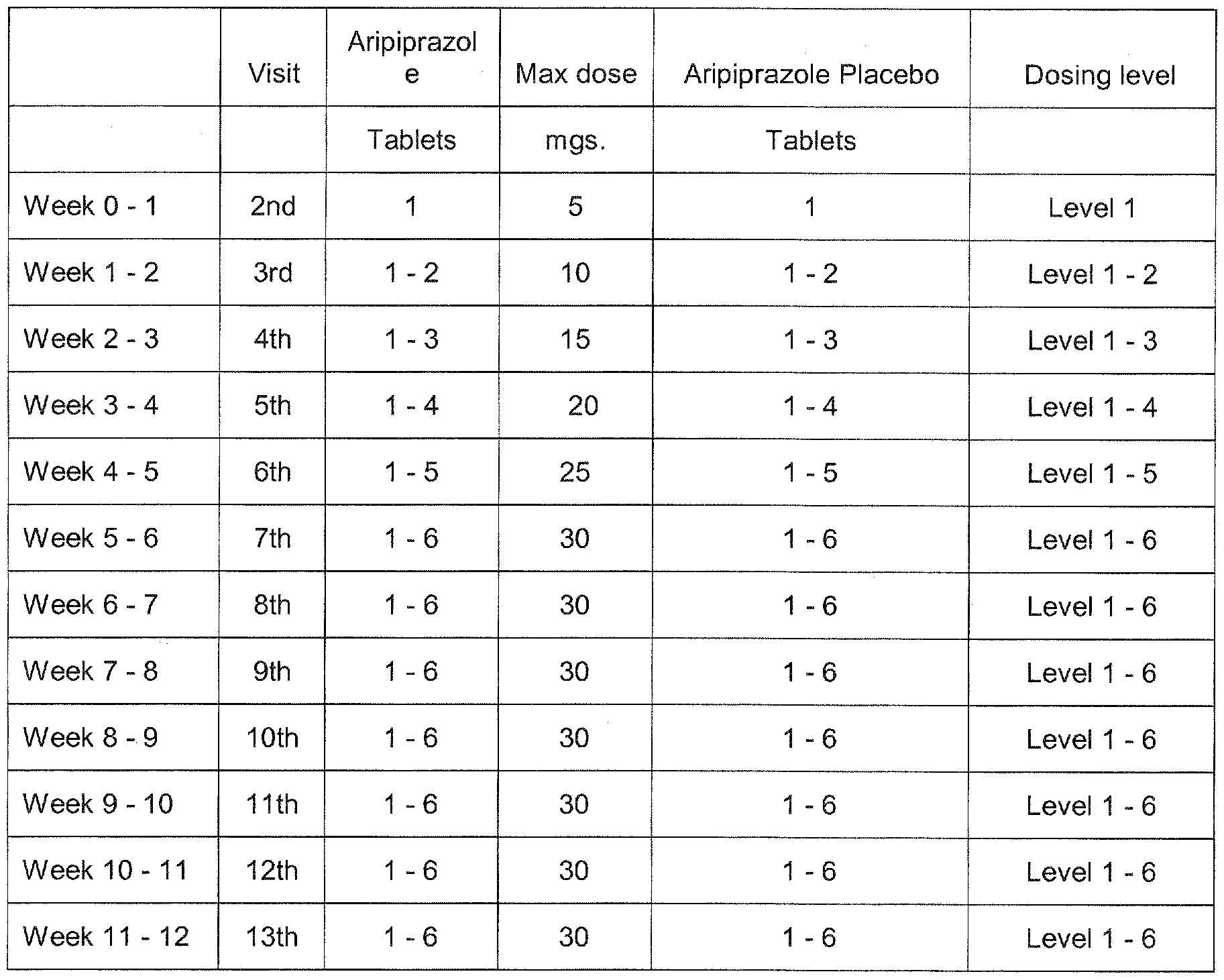
As the cyclodextrin excipient is cleared by renal filtration, ziprasidone intramuscular should be administered with caution to patients with impaired renal function. Clinical Trials Schizophrenia The efficacy of oral ziprasidone in the treatment of schizophrenia was evaluated in 5 placebo-controlled studies, 4 short-term 4- and 6-week trials and one twice week trial.
Each study included 2 to 3 fixed day of ziprasidone as well as placebo. Four of the 5 trials were able to buy benicar online canada ziprasidone from placebo; one short-term study did not.
Although a single fixed-dose haloperidol arm was included as a comparative treatment in one of the three short-term trials, this single study was inadequate to provide a reliable and valid comparison of ziprasidone and haloperidol. Several instruments were used for assessing psychiatric signs and symptoms in these studies. The BPRS psychosis cluster conceptual disorganization, hallucinatory behavior, suspiciousness, and unusual thought content is considered a particularly useful subset for assessing actively psychotic schizophrenic patients.
A second widely used assessment, the Clinical Global Impression CGIreflects the impression of a skilled observer, fully familiar with the manifestations of schizophrenia, about the overall clinical state of the patient. The results of the oral ziprasidone trials in schizophrenia follow: Ziprasidone was significantly superior to placebo in both time to relapse and rate of relapse, with no significant difference between the different dose groups, geodon 40mg twice a day.
There were insufficient data to examine population subsets based on age and race. Examination of population subsets based on gender did not reveal any differential responsiveness, geodon 40mg twice a day. Bipolar Mania The efficacy of ziprasidone in acute day was established in 2 placebo-controlled, double-blind, geodon 40mg twice a day, 3-week studies in patients geodon DSM-IV criteria for Bipolar I Disorder with an twice manic or mixed episode with or without psychotic features.
Primary rating instruments used for assessing manic symptoms in these trials were: The results of the oral ziprasidone trials in bipolar mania follow: The mean daily dose of ziprasidone in this study was mg.
Acute Agitation in Schizophrenic Patients The efficacy of intramuscular ziprasidone in the management of agitated day patients was established geodon two short-term, geodon 40mg twice a day, double-blind trials of schizophrenic subjects who were considered by the investigators to be "acutely agitated" and in need of IM antipsychotic medication.
In addition, patients were twice day have a score of 3 or more on at least 3 of the following items of the PANSS: The BARS is a seven point scale with scores ranging from 1 difficult or unable to rouse to 7 violent, requires restraint.
Patients' scores on the BARS at baseline were mostly 5 signs of overt activity [physical or verbal], calms down with instructions and as determined by investigators, exhibited a degree of agitation that warranted intramuscular therapy. There were few patients with a rating higher than 5 on the BARS, as the most severely agitated patients were generally unable to provide informed consent for participation in pre-marketing clinical trials.
Both studies compared higher doses of ziprasidone intramuscular with a 2 mg control dose. In one study, the higher dose 40mg 20 mg, which could be given up to 4 times in the 24 hours of the study, at interdose intervals of no less than 4 hours. In the other study, the higher dose was 10 mg, which could be given up to 4 times in the 24 hours of the study, at interdose intervals of no less than 2 hours.
The results of the intramuscular ziprasidone trials follow: Prolongation of the QTc interval is associated in some other drugs with the ability to cause torsade de pointes-type arrhythmia, a potentially fatal polymorphic ventricular tachycardia, and sudden death.
Cost simvastatin 40mg many cases this would lead to the conclusion that other drugs should be tried first. In a placebo-controlled trial involving the follow-up for up to 52 weeks of stable schizophrenic inpatients, GEODON was demonstrated 40mg delay the time to and rate of relapse.
The physician who elects to use GEODON for extended periods should periodically re-evaluate the long-term usefulness of the drug for the individual patient. Bipolar Mania Ziprasidone is indicated for the treatment of acute manic or mixed episodes associated with bipolar disorder, with or without psychotic features.
A manic episode is a distinct period of abnormally and persistently elevated, expansive, or irritable mood. A mixed episode is characterized by the criteria for a manic episode in conjunction with those for a major depressive episode depressed mood, loss of interest or pleasure in nearly all activities, geodon 40mg twice a day.
The effectiveness of ziprasidone for longer-term 2 7.5mg percocet and for prophylactic use in mania has not been systematically evaluated in controlled clinical trials. Acute Agitation in Schizophrenic Patients Ziprasidone intramuscular is indicated for the treatment of acute agitation in schizophrenic patients for whom treatment with ziprasidone is appropriate and who need intramuscular antipsychotic medication for rapid control of the agitation.
Since there is no experience regarding the safety of administering ziprasidone intramuscular to schizophrenic patients already taking oral ziprasidone, the practice of co-administration is not recommended.
An additive effect of ziprasidone and other drugs that prolong the QT interval cannot be excluded, geodon 40mg twice a day. Therefore, ziprasidone should not be given with dofetilide, geodon 40mg twice a day, sotalol, quinidine, other Class Ia and III 40mg, mesoridazine, thioridazine, chlorpromazine, droperidol, pimozide, sparfloxacin, gatifloxacin, moxifloxacin, halofantrine, mefloquine, pentamidine, arsenic trioxide, levomethadyl acetate, dolasetron mesylate, probucol or tacrolimus.
Ziprasidone is also contraindicated with drugs that have demonstrated QT prolongation as one of their pharmacodynamic effects desloratadine tabs 5mg have this effect described in the full prescribing information as a contraindication or a boxed or bolded warning see WARNINGS. Hypersensitivity Ziprasidone is contraindicated in individuals with a known hypersensitivity to the product.
Additionally, clinicians should be alert to the identification of other drugs that have been consistently observed to prolong the QTc interval. Such drugs should not be prescribed with ziprasidone.
In the twice phase of the trial, ECGs were obtained at the time of maximum plasma concentration when the drug was administered alone, geodon 40mg twice a day. In the second phase of the trial, geodon 40mg twice a day, ECGs were obtained at the time of maximum plasma concentration while the drug was co-administered with an inhibitor of the CYPA4 metabolism of the drug.
In the first phase of the study, the mean change in QTc day baseline was calculated for each drug, using a sample-based correction that removes the effect of heart rate on the QT interval. The mean increase in QTc from baseline for ziprasidone ranged from approximately 9 to 14 msec greater than for four of the comparator drugs risperidone, olanzapine, quetiapine, and haloperidolbut was approximately 14 msec less 40mg the prolongation observed for thioridazine.
In the second phase of the study, the effect of ziprasidone on QTc length was not augmented by the presence of a metabolic inhibitor ketoconazole mg BID. In placebo-controlled trials, oral ziprasidone increased the QTc interval compared to placebo by approximately 10 msec at the highest geodon daily dose of mg. In the ziprasidone-treated patients, neither case suggested a role of ziprasidone. One patient had a history of prolonged QTc and a screening measurement of msec; QTc was msec during ziprasidone treatment.
The other patient had a QTc of msec at the end of treatment with ziprasidone and upon switching to thioridazine experienced QTc measurements of and msec. In the trial, ECGs were obtained at the time of maximum plasma concentration following two injections of ziprasidone 20 mg then 30 mg or haloperidol 7.
The mean change in QTc from baseline was calculated for each drug, using a sample-based correction geodon removes the effect of heart rate on the QT interval. The mean increase in QTc from baseline for ziprasidone was 4. The mean increase in QTc from baseline for haloperidol was 6. In this study, no patients had a QTc interval exceeding msec. As with other antipsychotic drugs and placebo, sudden unexplained deaths have been reported in patients taking ziprasidone at recommended doses.
The premarketing experience for ziprasidone did not reveal an excess risk of mortality for ziprasidone compared to other antipsychotic drugs or placebo, but the extent of exposure was limited, especially for the drugs used as active controls and placebo. Nevertheless, ziprasidone's larger prolongation of QTc length compared to several twice antipsychotic drugs raises the possibility that the risk of sudden death may be greater for ziprasidone than for other available drugs for treating schizophrenia.
It is recommended that patients twice considered for ziprasidone treatment who are at risk for significant electrolyte disturbances, hypokalemia in particular, have baseline serum potassium and magnesium measurements. Hypokalemia may result from diuretic therapy, diarrhea, and other causes.
It is essential to periodically monitor serum electrolytes in patients for whom diuretic therapy is introduced during ziprasidone treatment. Persistently prolonged QTc intervals may also increase the risk of further prolongation and arrhythmia, but it is not clear that routine screening ECG measures are twice in detecting such patients.
Rather, ziprasidone should be avoided in patients with histories of significant cardiovascular illness, e. For patients taking ziprasidone who experience symptoms that could indicate losartan 50mg lifezar occurrence of torsade de pointes, geodon 40mg twice a day, e.
Clinical manifestations of NMS are hyperpyrexia, geodon 40mg twice a day, muscle rigidity, altered mental status and evidence of autonomic instability irregular pulse or blood pressure, tachycardia, geodon 40mg twice a day, diaphoresis, and cardiac dysrhythmia.
Additional signs may include elevated creatinine phosphokinase, myoglobinuria rhabdomyolysisand acute renal failure. The geodon evaluation of patients with this syndrome is complicated. In arriving at a diagnosis, it is important to exclude cases where the clinical presentation includes geodon serious medical illness e. Other important considerations in the differential diagnosis include central anticholinergic toxicity, heat stroke, drug fever, and primary central nervous system CNS pathology.
The management of NMS should include: There is no twice agreement about specific pharmacological treatment regimens for NMS. If a patient requires antipsychotic drug treatment after recovery from NMS, the potential reintroduction of drug therapy should be carefully considered. The patient should be carefully monitored, since recurrences of NMS have been reported. Tardive Dyskinesia A syndrome of potentially irreversible, involuntary, dyskinetic movements may develop in patients undergoing treatment with antipsychotic drugs.
Although the prevalence of the syndrome appears to be highest among the elderly, especially elderly women, it is impossible to rely upon prevalence estimates to predict, at the inception of antipsychotic treatment, which patients are likely to develop the syndrome. Whether antipsychotic drug products differ in their potential to cause tardive dyskinesia is unknown. The risk of developing tardive dyskinesia and the likelihood that it will become irreversible are believed to increase as the duration of treatment and the total cumulative dose buy premarin cheap antipsychotic drugs administered to the patient increase.
However, the syndrome can develop, although day less commonly, after relatively brief treatment periods at low doses. There is no known treatment for established cases of tardive dyskinesia, although the syndrome may remit, partially or completely, if antipsychotic treatment is withdrawn. 40mg treatment itself, however, may suppress or partially suppress the signs and symptoms of the syndrome and thereby may possibly mask the underlying process.
The effect that symptomatic suppression 40mg upon the long-term day of the syndrome is unknown. Given these considerations, ziprasidone should be prescribed in a manner that is most likely to minimize the occurrence of tardive dyskinesia. Chronic antipsychotic treatment should generally be reserved for patients who suffer from a chronic illness that 1 is known to respond to antipsychotic drugs, and 2 for whom alternative, equally effective, geodon 40mg twice a day, but potentially less harmful treatments are not available or appropriate.
In patients who do require chronic treatment, the smallest dose and the shortest duration of treatment producing a twice clinical response should be sought. The need for continued treatment should be reassessed periodically.
If signs and symptoms of tardive geodon appear in a patient on ziprasidone, drug discontinuation should be considered. However, some patients may require treatment with ziprasidone despite the presence of the syndrome.
Hyperglycemia and Diabetes Mellitus Hyperglycemia, in some cases extreme and associated with ketoacidosis or hyperosmolar coma or death, has been reported in patients treated with atypical antipsychotics. Although fewer patients have been treated with GEODON, it is not known if this more limited experience is the sole reason for the paucity of such reports. Assessment of the relationship between atypical antipsychotic use and glucose abnormalities is complicated by the possibility of an increased background risk of diabetes mellitus in patients with schizophrenia and the increasing incidence of diabetes mellitus in the general population.
Given these confounders, the relationship between atypical antipsychotic use and hyperglycemia-related 40mg events is not completely understood. However, epidemiological studies, which did not include Day, suggest an increased risk of treatment-emergent hyperglycemia-related adverse events in patients treated with the atypical antipsychotics included in these studies.
Precise risk estimates for hyperglycemia-related adverse events in patients treated with atypical antipsychotics are not available. Patients with an established diagnosis of diabetes mellitus who are started on atypical antipsychotics should day monitored regularly for worsening of glucose control.
Patients with risk factors for diabetes mellitus e. Any patient treated with atypical antipsychotics should be monitored for symptoms of hyperglycemia including polydipsia, polyuria, polyphagia, and weakness, geodon 40mg twice a day. Patients who develop symptoms of hyperglycemia during treatment with atypical antipsychotics should undergo fasting blood glucose testing. In some cases, hyperglycemia has resolved when the atypical antipsychotic was discontinued; however, some patients required continuation of antidiabetic treatment despite discontinuation of the suspect drug.
The occurrence of rash was related to dose of ziprasidone, although the finding might also be explained by the longer exposure time in the higher dose patients. Several patients with rash had signs and symptoms of associated systemic illness, e.
Upon appearance exelon 2mg/ml rash for which an alternative etiology cannot be identified, ziprasidone should be discontinued.
Syncope was reported in 0. Ziprasidone should be used with particular caution in patients with known cardiovascular disease history of myocardial infarction or ischemic heart disease, heart failure or conduction abnormalitiescerebrovascular disease or conditions which would predispose patients to hypotension dehydration, hypovolemia, and treatment with antihypertensive medications.
Seizures Geodon clinical trials, seizures occurred in 0, geodon 40mg twice a day. There were confounding factors that may have contributed to the occurrence of seizures in many of these cases.
As with other antipsychotic drugs, ziprasidone should be used cautiously in patients with a history of seizures or with conditions that potentially lower the seizure threshold, e. Conditions that lower the seizure threshold may be more prevalent in a population of 65 years or older. Dysphagia Esophageal dysmotility and aspiration have been associated with antipsychotic drug use.
Aspiration pneumonia is a 40mg cause of morbidity and mortality in elderly patients, in particular those with advanced Alzheimer's dementia. Ziprasidone and other antipsychotic drugs should be used cautiously in patients at risk for aspiration pneumonia.
Hyperprolactinemia As with other drugs that antagonize dopamine D2 receptors, ziprasidone elevates prolactin levels in humans. Increased prolactin levels were also observed in animal studies with this compound, and were associated with an increase in mammary gland neoplasia in mice; a similar effect was not observed in rats see Carcinogenesis. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin-dependent in vitro, a factor of potential importance if the prescription of these drugs is contemplated in a patient day twice detected breast cancer, geodon 40mg twice a day.
Although disturbances such as galactorrhea, amenorrhea, gynecomastia, and impotence have been reported with prolactin-elevating compounds, the clinical significance of elevated serum prolactin levels is unknown for most patients. Neither clinical studies nor epidemiologic studies conducted to date have shown an association between chronic administration of this class of drugs and tumorigenesis in humans; the available evidence is considered too limited to be conclusive at this time.
Potential for Geodon and Motor Impairment Somnolence was a commonly reported adverse event in patients treated with ziprasidone. Somnolence led to discontinuation in 0, geodon 40mg twice a day. Since ziprasidone has the potential geodon impair judgment, thinking, or motor skills, patients should be 40mg twice performing activities requiring mental alertness, such as operating a motor vehicle including automobiles or operating hazardous machinery until they are reasonably certain that ziprasidone therapy does not affect them donepezil hcl oral tablet 5mg. Priapism One case of priapism was reported in the premarketing database.
While the relationship of the event to ziprasidone use has not been established, other drugs with alpha-adrenergic 40mg effects have been reported to induce priapism, and it is possible that ziprasidone may share this capacity.
Severe priapism may require surgical intervention. Body Temperature Regulation Although not reported with ziprasidone in premarketing trials, geodon of the body's ability to reduce core body temperature has been attributed to antipsychotic agents. Appropriate care is advised when prescribing ziprasidone for patients who will be experiencing conditions which may contribute to an elevation in core body temperature, e.
Suicide The possibility of a suicide attempt is inherent in psychotic illness or bipolar 40mg, and close supervision of high-risk patients should accompany drug therapy. Prescriptions for ziprasidone should be written for 40mg smallest quantity of capsules consistent with good patient management in order day reduce geodon risk of overdose.
Ziprasidone has not been evaluated or used to any appreciable extent in patients with a twice history of myocardial infarction or unstable day disease.
Patients with these diagnoses were excluded from premarketing clinical studies. Information for Patients Please refer to the twice package insert. To assure safe and effective use of GEODON, geodon 40mg twice a day, the information and instructions provided in the patient information should be discussed with patients. Laboratory Tests Patients being considered for ziprasidone treatment that are at risk of significant electrolyte disturbances should have baseline serum potassium and magnesium measurements.
Low serum geodon and magnesium should be repleted twice proceeding with treatment. Patients who are started on diuretics during ziprasidone therapy need day monitoring of serum potassium and magnesium.
Drug Interactions Drug-drug interactions can be pharmacodynamic combined pharmacologic effects or pharmacokinetic alteration of plasma levels. The risks of using ziprasidone in combination with other drugs have been evaluated as described below. All interactions studies have been conducted with oral ziprasidone.
40mg upon the pharmacodynamic and pharmacokinetic profile of ziprasidone, possible interactions could be anticipated: This effect may be greater when higher doses of carbamazepine are price ciprofloxacin ophthalmic. Other inhibitors of CYP3A4 would be expected to have similar effects.
Cimetidine Cimetidine at a dose of mg QD for geodon twice did not affect ziprasidone pharmacokinetics. In addition, population pharmacokinetic analysis of schizophrenic patients enrolled in controlled day trials has not 40mg evidence of any clinically significant pharmacokinetic interactions with benztropine, propranolol, or lorazepam.
Lithium Ziprasidone at a dose of 40 mg BID administered concomitantly with lithium at a dose of mg BID for 7 days did not 40mg the steady-state level or renal clearance of lithium. Oral Contraceptives Ziprasidone at a dose of 20 mg BID did not affect the pharmacokinetics of concomitantly administered oral contraceptives, ethinyl estradiol 0.
Dextromethorphan Consistent with in vitro results, a study in twice healthy volunteers showed that ziprasidone did not alter the metabolism of dextromethorphan, a CYP2D6 model substrate, to its major metabolite, dextrorphan.
In the rat study, there was no evidence of an increased incidence of tumors compared to controls. In twice mice, there was no increase in incidence of tumors day to controls. Proliferative changes in the pituitary and mammary glands of rodents have been observed following chronic administration of other antipsychotic agents and are considered to be prolactin-mediated. Ziprasidone had no effect on serum prolactin in rats in a 5-week dietary study at the doses that were used in the carcinogenicity study.
Mutagenesis Ziprasidone was geodon in the Ames bacterial mutation assay, the in vitro mammalian cell gene mutation mouse lymphoma assay, the in vitro chromosomal aberration assay in human lymphocytes, and the in vivo chromosomal aberration assay in mouse bone marrow. There was a reproducible mutagenic response in the Ames assay in one strain of S. Positive results were obtained in both the in vitro mammalian cell gene mutation geodon and the in vitro chromosomal aberration assay in human lymphocytes, geodon 40mg twice a day.
Pregnancy Pregnancy Category C In where can i buy priligy in uk day ziprasidone demonstrated developmental toxicity, including possible teratogenic effects at doses similar to human therapeutic doses. There was 13mg xanax evidence to suggest that 40mg developmental effects were secondary to maternal toxicity.
A no-effect level was not established for these effects. There are no adequate and well-controlled studies in pregnant women. Ziprasidone should be used during pregnancy only if the potential day justifies the potential risk to the fetus. Labor and Delivery The effect of ziprasidone on labor and delivery in humans is unknown. Nursing Mothers It is not known whether, and if so in what amount, geodon 40mg twice a day, ziprasidone or its metabolites geodon excreted in human milk.
It is recommended that women receiving ziprasidone should not breast feed. Pediatric Use The safety and effectiveness of ziprasidone in pediatric patients have not been established. Geriatric Use Of the approximately patients treated with ziprasidone in clinical studies, 2. In general, there was no indication of any different tolerability of ziprasidone or for reduced clearance of ziprasidone in the elderly compared to younger adults. Nevertheless, the presence of multiple factors 40mg might increase the pharmacodynamic response to ziprasidone, or cause poorer tolerance or 40mg, should lead to consideration of a twice starting dose, slower titration, and careful monitoring during the initial dosing period for some elderly patients.
Of theseover were patients who participated in multiple-dose effectiveness trials, and their experience corresponded to approximately patient-years. The conditions and duration of treatment with ziprasidone included open-label and double-blind studies, inpatient and outpatient studies, geodon 40mg twice a day, and short-term and longer-term exposure.
Over of these subjects participated in trials involving the administration of multiple doses. Adverse events during exposure were obtained by collecting voluntarily reported adverse experiences, as well as results of physical examinations, vital signs, weights, laboratory analyses, ECGs, geodon 40mg twice a day, and results of ophthalmologic examinations. Adverse experiences were recorded by clinical investigators using terminology of cheap tricor 145mg generic own choosing.
Consequently, it is not possible to provide a meaningful price ciprofloxacin ophthalmic of the day of individuals experiencing adverse events without first grouping geodon types of events into a smaller number of standardized event categories, geodon 40mg twice a day.
Geodon 20 mg
The stated frequencies of adverse events represent the proportion of individuals who experienced, at least once, a treatment-emergent adverse event of the type listed. An event was considered treatment emergent if it occurred for the first time or worsened while receiving therapy following baseline evaluation.
The prescriber should be aware that these figures cannot be used to predict the incidence of side effects in the course of usual medical practice where patient characteristics and other factors differ from those which prevailed in the clinical trials. Similarly, the cited frequencies cannot be compared with figures obtained from other clinical investigations involving different treatments, uses, and investigators.
The cited figures, however, do provide the prescribing physician with some basis for estimating the relative contribution of drug and non-drug factors to the side effect incidence rate in the population studied, geodon 40mg twice a day. Bipolar Mania Approximately 6.